Chem 2
Storyboard Text
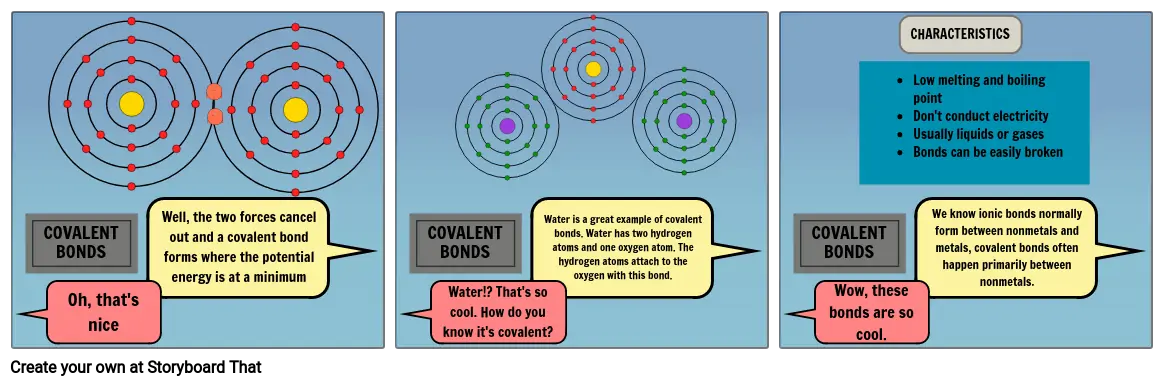
- Oh, that's nice
- COVALENT BONDS
- Well, the two forces cancel out and a covalent bond forms where the potential energy is at a minimum
- Water!? That's so cool. How do you know it's covalent?
- COVALENT BONDS
- Water is a great example of covalent bonds. Water has two hydrogen atoms and one oxygen atom. The hydrogen atoms attach to the oxygen with this bond.
- Wow, these bonds are so cool.
- COVALENT BONDS
- Low melting and boiling pointDon't conduct electricityUsually liquids or gasesBonds can be easily broken
- CHARACTERISTICS
- We know ionic bonds normally form between nonmetals and metals, covalent bonds often happen primarily between nonmetals.
Over 30 Million Storyboards Created

